Breast cancer pathology relies on multiple immunohistochemistry biomarkers, each requiring accurate and consistent interpretation to support diagnosis and treatment decisions. As case volumes increase and biomarker menus expand, pathology teams face growing pressure to maintain standardization and efficiency, often across multiple readers, sites, and workflows. Fragmented digital tools and manual interpretation can introduce variability, limit scalability, and increase operational burden.
HistoSiA™ Breast Cancer Suite by OptraSCAN® is a digital pathology solution that supports algorithm-assisted quantitative interpretation of key breast cancer biomarkers on a unified workflow.
Powered by the world’s first patented OS-SiA™ technology, HistoSiA™ Breast combines whole-slide imaging with real-time AI analysis, allowing laboratories to scan slides, automatically analyze biomarker expression, and segregate breast cancer IHC cases into diagnostic* categories in a single workflow before the pathologist even begins review. This innovative multi-biomarker expansion platform allows integration of HER2, ER, PR, Ki-67, and AE1/3 biomarker analyses within the same digital workflow.
The platform performs automated HER2 categorization into 0, 1+, 2+, and 3+ classes during slide scanning, enabling laboratories to triage cases, reduce manual scoring workload, and streamline pathologist review.
This is an innovative multi-biomarker expansion platform that allows scalable integration of additional breast cancer biomarker analyses, such as ER, PR, Ki-67, and AE1/3, within the same digital workflow.
Scan:
Slides are prepared using standard laboratory protocols and processed on OptraSCAN systems (OS-SiX™, OS-Lite™,OS-Ultra™, and OS-FLi™) to generate high-resolution digital slide images for analysis.
Index:
Automatically generates indexed thumbnails, slide maps, metadata,and contextual information. Once the scan is complete, the slides are already categorized, cataloged, and made searchable. Each scanned region is instantly tagged, and the database is prepared for easy integration into the lab workflow.
Analyze:
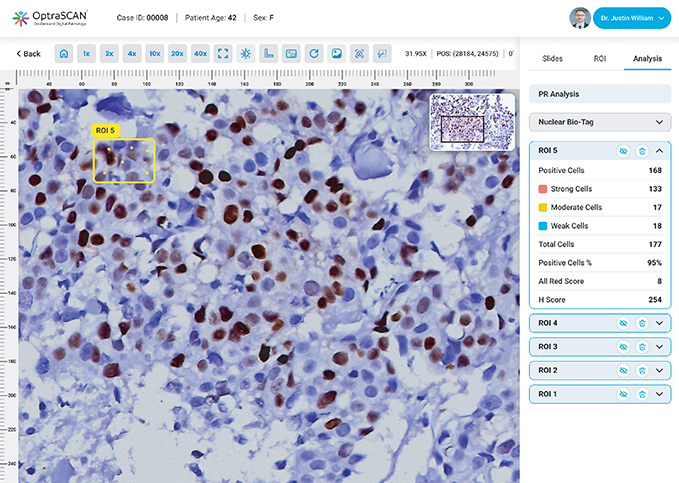
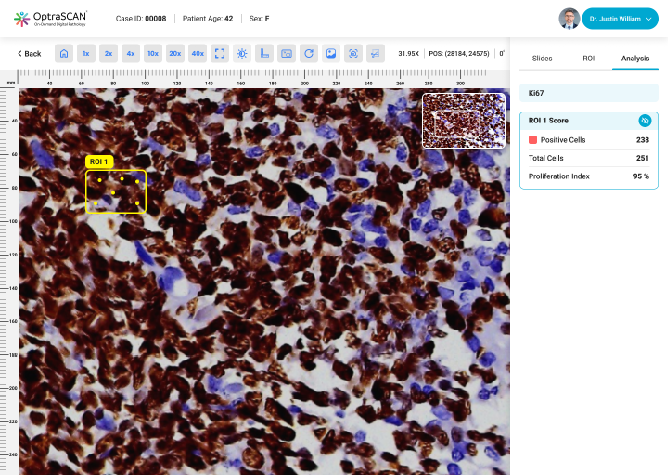
During acquisition, HistoSiA AI generates IHC quantitative analysis across entire tissue sections. It triages slides as negative or positive and displays visual overlays and numerical outputs to support interpretation and help prioritize pathologist's review. Confirmed positive smears are graded using built-in scoring tools e.g., Allred, H-Score (ER, PR), ASCO-CAP (HER2), Proliferation index (Ki-67), TNM staging. All images and reports are managed digitally in IMAGEPath® software.
HistoSiA™ Breast delivers interpretable analytical results designed for real-world pathology workflows
Quantitative analysis includes:
(Section intended to display representative outputs for HER2, ER, PR, and Ki-67)
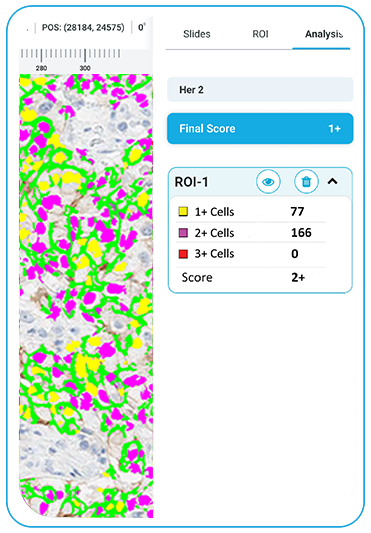
HER2
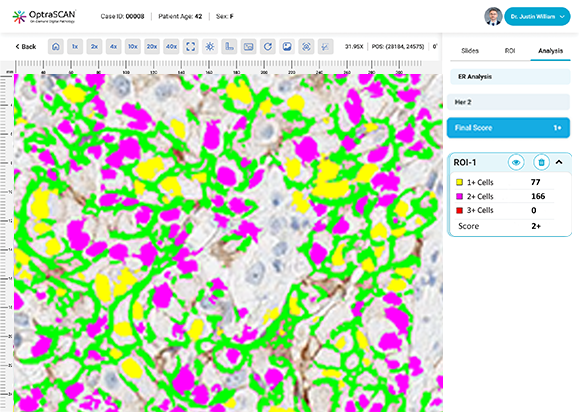
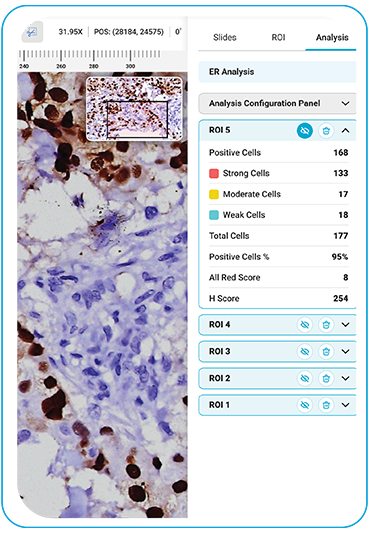
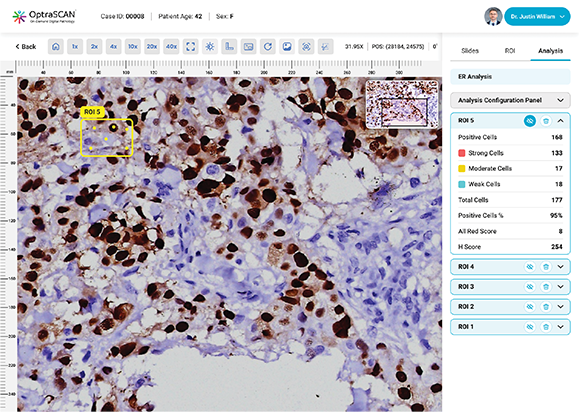
ER
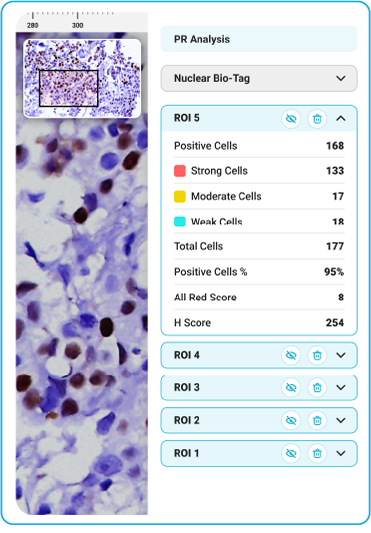
PR
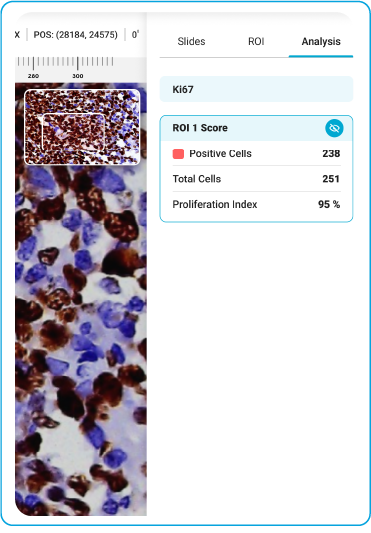
Ki-67
Scalable platform architecture
Supports quantitative pathology across multiple disease areas
Enables labs to expand capabilities over time
Eliminates need to reinvest in new systems or workflows
Enables quantitative and semi-quantitative IHC analysis in breast pathology
Aligns with existing CPT® coding for quantitative IHC
Includes CPT® 88361 – Per specimen, per antibody
Coding, coverage, and reimbursement vary by payer and institution

Please fill out the form below and complete all questions with an asterisk (*)